Amplatzer™ Valvular Plug III
Paravalvular leaks (PVLs) are a common and challenging problem around the world.1 The Amplatzer™ Valvular Plug III is specially designed to provide an effective solution to this key issue, improving quality of life and longevity for an increasing number of patients.2-5
DELIVERING AN EFFECTIVE SOLUTION FOR A WIDE RANGE OF PVL MORPHOLOGIES2,6,7
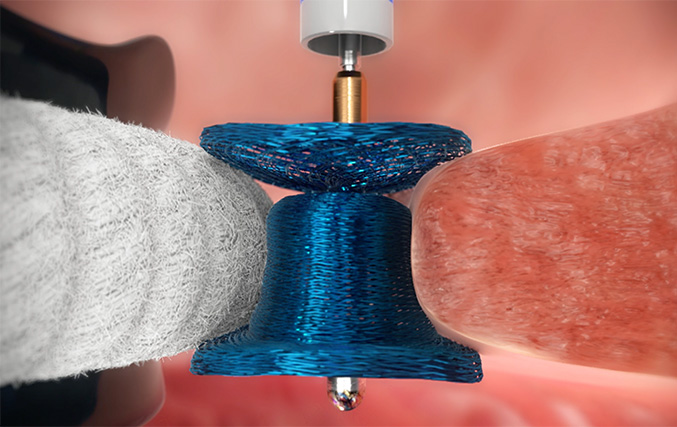
Paravalvular leaks (PVL) can be found in a variety of shapes and sizes. To tackle the leaks and fill the gaps of different morphologies, the Amplatzer™ Valvular Plug III is designed in an asymmetrical oblong shape that fits a wide range of PVL. As such, the Amplatzer Valvular Plug III can provide effective closure in up to 93% of patients.1, 5, 6
PATIENTS WHO BENEFIT
A solution to a key issue
Around the world, PVL are a common and challenging problem, occurring in 7% to 17% of mitral valve replacements (MVR) and 5% to 10% of aortic valve replacements (AVR).1 Risk factors for PVL development include: annular calcification, tissue friability, prior endocarditis, or other inflammatory processes, and recent initiation of corticosteroid therapy.8 PVL can occur soon after valve replacement or several years later.2.6
By providing an effective solution to this key issue, the Amplatzer™ Valvular Plug III is improving quality of life for an increasing number of patients.2-5
KEY FEATURES
Built on the extensive Amplatzer™ legacy of safety and efficacy
- Pioneered transcatheter cardiovascular and peripheral vascular occlusion
- Over 1.25 million Amplatzer devices implanted worldwide9
- More than 20 years of clinical experience and global leadership
Every detail is designed for successful PVL closure
The Amplatzer Valvular Plug III has been specially designed to offer patients an effective solution to PVL. This oblong, self-expanding device is made from braided Nitinol wires with shape memory characteristics, making it well suited to PVL closure.2,6,7,9,10
To close PVL with different morphologies, the Amplatzer Valvular Plug III comes in a range of 9 sizes and has a flexible Nitinol waist, ensuring an excellent device-to-patient fit.

MAT-2201627 v4.0 | Item approved for OUS use only.