PDA Closure
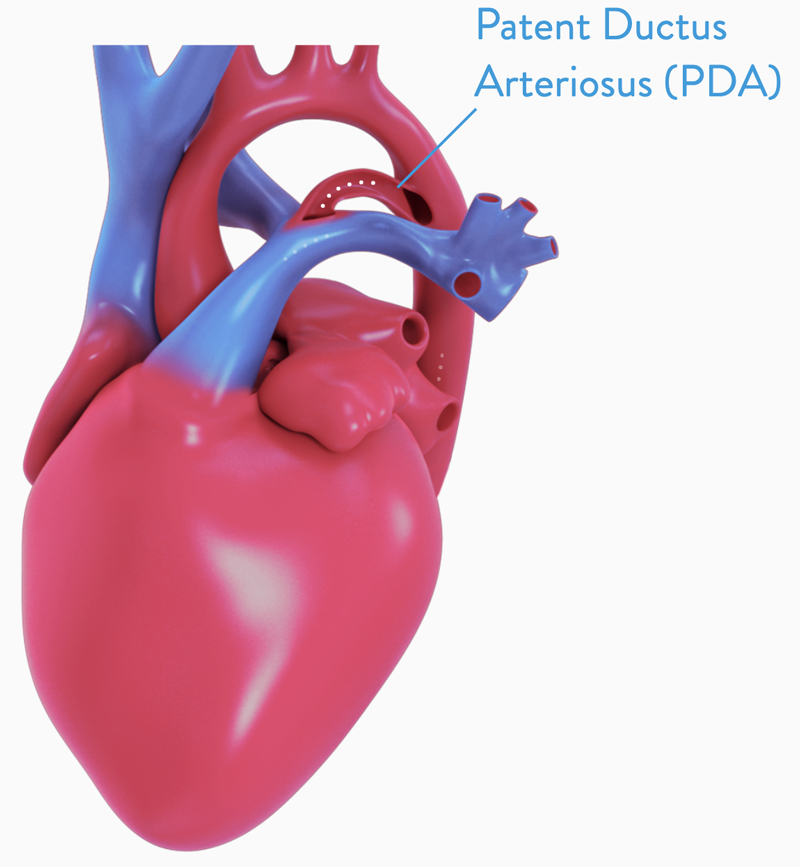
PATENT DUCTUS ARTERIOSUS
TREATMENT OPTIONS
There are a number of treatment options for a PDA, and there is no single option that is right for every person. You should talk with your doctor to learn about the best treatment option for you or your child; however, there are a few standard approaches of which you should be aware.
The first option is medication, which may be appropriate to help close the PDA or in treating symptoms associated with the PDA.
Other treatment options include open-heart surgery or a minimally-invasive catheter-based procedure, like the Amplatzer™ Duct Occluder.
DISCUSS ALL TREATMENT OPTIONS WITH YOUR DOCTOR
Your doctor can describe the risks and benefits and help you decide which option is right for you.
The Amplatzer™ Duct Occluder,
Duct Occluder II & Piccolo™ Occluder
The Amplatzer Duct Occluders are designed for nonsurgical PDA closure. The devices have different design features, and your doctor will select the most appropriate device for your PDA. The device is placed in the PDA during a catheter-based procedure and will remain permanently implanted.
Each device is made from braided nitinol wires. Nitinol is a metal with shape memory characteristics, meaning the device will return to its original shape even after it is stretched to pass through a catheter. The shape of each device was specifically designed to stop blood flow through a PDA.

Frequently asked questions
- How do I know which treatment option is right for me?
Every person is unique. Your doctor is your best resource for learning about the treatment options available to you and the best course for your condition. Talk to your doctor and follow his or her advice for your care. Remember a PDA can result in unpleasant symptoms and increased health risk. With proper care, however, it can generally be managed with medication or closure.
- What is involved with a catheter-based procedure?
A catheter-based procedure is a minimally invasive treatment option available to some people. The procedure involves making a small incision, typically in the groin, and inserting a small tube, called a catheter, to navigate through the blood vessels to the procedure site within the heart.
In people with a PDA, the doctor guides the device through the catheter to seal the PDA. Once the device is placed in the PDA, the doctor will carefully study its position using cardiac imaging systems. Once satisfied with the position, the device is released to remain permanently in the channel. The catheter is removed and the procedure is completed.
The procedure itself should last about one to two hours and will take place in a heart catheterization laboratory, where many minimally invasive, nonsurgical procedures are performed. Your doctor may give you an anesthetic, and you should not feel any significant discomfort.
- Who should not receive an Amplatzer Duct Occluder or Duct Occluder II?
If you have any of the following conditions, you may not be a good candidate to receive the Amplatzer Duct Occluder.
- If you weigh less than 6 kg.
- If you are less than 6 months of age.
- If you have blood clots in their heart or vessels.
- If you have an infection.
- If have blood clots in your heart.
- If your heart or your veins are too small or if they cannot undergo the procedure.
- If your heart or your veins are very small or if there is any reason you cannot undergo the procedure.
- If you have high blood pressure in the pulmonary arteries.
If you have any of the above conditions or any of the following conditions, you may not be a good candidate to receive the Amplatzer Duct Occluder II.- If you have a window-type PDA.
- If your blood flows from right to left through the PDA.
- If you have other heart defects that require surgery.
- If you have had more than two lower respiratory infections within the past year.
- If you are unable to take blood thinner medication.
- If your heart or your veins are very small or if there is any reason you cannot undergo the procedure.
- If the size of your ductus (channel) is too large for the device.
- Who should not receive the Amplatzer Piccolo Occluder?
If your child has any of the following conditions, they may not be a good candidate to receive the Amplatzer Piccolo Occluder.
- If your child weighs less than 700 grams at the time of the procedure
- If your child is fewer than 3 days old at the time of the procedure
- If your child has a narrowing in the aorta or in the left pulmonary artery
- If your child has high blood pressure in the pulmonary arteries and the blood flow to the rest of the body must go through the PDA
- If your child's PDA length is shorter than 3mm
- If your child's PDA diameter at the narrowest portion is greater than 4mm
- If your child has an active infection
- If your child has a clot (thrombus) in their heart that may interfere with the implant procedure
- Will medical equipment interfere with my device?
Although most medical equipment will have no effect on your device, it is best to tell hospital personnel that you have an implanted device before you undergo any medical procedure. Magnetic resonance imaging (MRI) scans are generally acceptable, and your Amplatzer occlusion device has no known hazards when using a 3-tesla MRI, an MRI system more powerful and faster than standard MRI machines. If an MRI is needed, simply inform the MRI staff about your implant.
- What happens after the procedure?
Because the procedure is minimally invasive, recovery will likely be quick and easy. In the absence of other medical conditions that require hospitalization, many people are discharged from the hospital within 24 hours. Your doctor can provide guidelines for activities and medications. He or she will prescribe drugs that you should take at home to continue your treatment and recovery. The decision to prescribe these is at the discretion of your doctor. Many doctors require follow-up appointments over the next year to ensure recovery is going well. What to expect during and after the procedure will vary. Discuss all questions and concerns you have with your doctor.
- How long will it take me to recover, what activities should be avoided after the procedure, and when can they resume?
Every person recovers differently, and your doctor can help determine when activities can be resumed. In general, all strenuous activity should be avoided for one month after the procedure.
- Will I be able to feel the device?
No, you will not be able to feel the device once it’s implanted.
- Can I travel with an implanted device? Will my device trigger airport security systems?
Your physician is your best resource for the answer to this question. Many people find that with some extra planning and care they can enjoy traveling even with an implanted device. It is always wise to carry your patient ID card, just in case you encounter difficulties while traveling.
Though some people worry about airport security systems there is really no need for concern. The metal parts in Amplatzer occlusion devices are very small and usually do not trigger metal detector alarms. However, the sensitivity setting of the metal detector and other factors may affect how the metal detector responds to your device. Simply show your patient identification card to security personnel.
- What is a patient identification card? Will I need to carry it with me?
As a device recipient, it is important to carry a patient identification card with you to identify yourself as having an implanted device. The patient ID card includes your name, implant date, your doctor’s contact information and information about your device. You will be provided with this card after the procedure.
- Can I have this procedure if I am pregnant? What if I am a nursing mother?
The risk of increased x-ray exposure must be weighed against the potential benefits of this device. Your physician will ensure that care will be taken to minimize the radiation exposure to the fetus and mother.
It is unknown if the device affects breast milk. You should discuss this issue with your doctor.
- After the procedure, what symptoms should I seek medical help for?
If you experience any of following symptoms after the procedure: chest pain, numbness, sudden weakness, dizziness or rapid heartbeat, seek medical help immediately. An echocardiogram (ultrasound of the heart) should be performed.
- What risks are associated with the Amplatzer Duct Occluder and Duct Occluder II?
There are certain potential risks associated with catheter-based procedures as well as additional risks that may be associated with the device. Your doctor is the best source of information about the risks of having an implanted device. Be sure to talk about all your questions and concerns.
Potential risks include, but are not limited to:
- Air embolus (an air bubble that blocks blood flow in a vessel)
- Allergic drug reaction
- Allergic dye reaction
- Anesthesia reaction
- Apnea (temporary absence of breathing)
- Arrhythmia (loss of regular heart rhythm)
- Bacterial endocarditis (infection that causes swelling of the lining of the heart and its valves)
- Bleeding
- Brachial plexus injury (injury to the nerves in the arm or lower neck)
- Chest pain
- Death*
- Delivery system failure
- Fever
- Headache/Migraine
- Hyper/Hypotension (abnormally high/low blood pressure)
- Myocardial infarction (heart attack)
- Partial obstruction of pulmonary artery
- Perforation of vessel or myocardium (piercing of a vessel or the heart)
- Peripheral embolism (when a small clot or piece of debris passes through the peripheral system causing decreased or blocked blood flow in an artery or vein)
- Stroke/Transient ischemic attack (temporary lack of oxygen to the brain)
- Thrombus (blood clot)
- Valvular regurgitation or insufficiency (abnormal backward flow of blood through a valve)
- Vascular access site complications
You should also be aware that:- The Amplatzer Duct Occluder contains nickel-titanium alloy, which is generally considered safe. However, in vitro testing has demonstrated that nickel is released from this device for a minimum of 60 days. Patients who are allergic to nickel may have an allergic reaction to this device, especially those with a history of metal allergies. Certain allergic reactions can be serious; patients should be instructed to seek medical assistance immediately if they suspect they are experiencing an allergic reaction. Symptoms may include difficulty in breathing or swelling of the face or throat. While data are currently limited, it is possible that some patients may develop an allergy to nickel if this device is implanted.
- Patients allergic to nickel may suffer an allergic reaction to this device.
- There is limited clinical data for patients over 40 years of age.
- If you are pregnant, you and your baby are at risk for increased X-ray exposure. Notify your doctor if you are (or believe you might be) pregnant.
- If the device were to be dislodged, you may need surgery for its removal. Your PDA will be repaired at the same time. Surgery following device placement may be more difficult.
For additional information, please contact your doctor.
The information provided is not intended for medical diagnosis or treatment or as a substitute for professional advice. Consult with a physician or qualified healthcare provider for appropriate medical advice.
MAT-2305397 v2.0 | Item approved for U.S. use only.